The performance and bio-integration of the OSSIOfiber® Intelligent Bone Regeneration Technology has been successfully evaluated for in-bone and soft tissue fixation. The purpose of this in vivo study was to further evaluate the OSSIOfiber® material properties when implanted over the surface of bone, for its bone-implant interface, local tissue response and full bio-integration profile. This study serves as an indicator for the potential of OSSIOfiber as plate fixation for trauma, extremities and pediatrics.
Test Device: OSSIOfiber Plates*
- 50mmx10mmx2mm plates comprised of OSSIOfiber Intelligent Bone Regeneration Technology with 4 pre-drilled screw holes
- At the time, OSSIOfiber screws were not readily available for use, thus metal (stainless-steel) cortical screws were utilized to fixate the OSSIOfiber plates onto bone
*Note: OSSIOfiber Plates are not available for sale
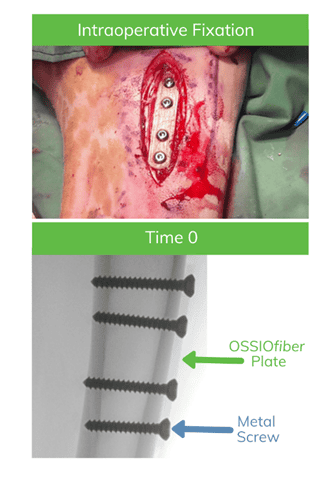
Methods:
- Test model: 8 mature female sheep, average weight 70kg
- Medial approach to the proximal tibia: skin incision followed by blunt dissection down to the bone surface
- OSSIOfiber plates were placed approximately 3cm distal to the tibial plateau, parallel to the tibial tuberosity and directly on the cortical bone surface, using bi-cortical fixation with four metal screws
- Standard wound closure was performed
- Animals were allowed to fully load bear immediately following surgery and were closely monitored for any adverse clinical signs
- Micro-CT and histopathology evaluation were conducted at 13, 26, 52, 78, and 104 weeks

At 104 weeks, Micro-CT demonstrates that the OSSIOfiber plates to be essentially fully integrated into the native anatomy.
Results
- Quiescent, gradual elimination of the OSSIOfiber plates through 104 weeks of implantation
- No adverse clinical observations or local bone/soft tissue reactions (i.e. local swelling, cyst formation, lameness) were noted through 104 weeks of implantation
- At 104 weeks, Micro-CT demonstrates the OSSIOfiber plates to be essentially fully integrated into the native anatomy
Conclusion
OSSIOfiber Intelligent Bone Regeneration Technology, as a plate construct fixated on bone, demonstrated the ideal bio-integration profile for a plate fixation device. Gradual integration, restoring the native anatomy without adverse foreign body reactions, allowing the bone to return to the pre-injury state without forming a permanent callus or overgrowth of new bone.
The development of OSSIOfiber screws will enable new plate and screw system solutions that are fully bio-integrative and avoid the costs, risks and burdens of permanent metal hardware removal. Through the engineering and manufacturing capabilities at OSSIO, plate and screw systems comprised of strong and bio-integrative OSSIOfiber can be designed for optimal biomechanical capabilities and for a multitude of fixation applications across trauma, extremities and pediatrics. OSSIO is actively developing and building evidence for OSSIOfiber plate and screw systems.
Data on File at OSSIO.
OSSIOfiber Compression Plates are not yet available for sale. OSSIOfiber Compression Screws are not available for sale outside of the US.
® OSSIO and OSSIOfiber are registered trademarks of OSSIO Ltd. All rights reserved.
DOC0001433 Rev 01 12/2020
